Exploring the Challenges & Opportunities for Label-free Optical Imaging
Marcia Lesky
Their objective in organizing this program was to develop vision and strategies for accelerating the translation of label-free optical techniques for clinical practice. They hope to do that through identifying opportunities for label-free modalities, identifying barriers to clinical translation and proposing solutions.

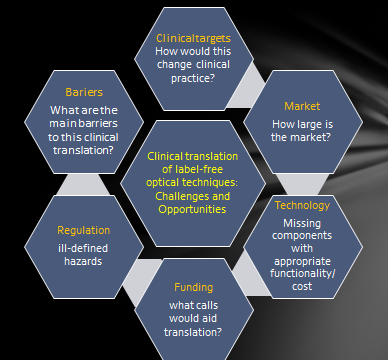
Despite their inherent advantages, the translation of label-free optical technologies to clinical settings has not been widely adopted in medical practice. New paradigms for diagnosis and clinical management of health (e.g. improved diagnosis, real-time guidance of interventions/surgery, monitoring of the efficacy of therapy, and brain optical imaging) are clearly needed. Rapid label-free diagnosis of diseases in vivo can enable development of new clinical devices addressing a broad range of disorders including many cancers, cardiovascular, ophthalmologic and neurological diseases.
The participants have already begun to identify and evaluate the main bottlenecks for clinical translation of label-free optical techniques including technological and regulatory challenges. This afternoon and tomorrow they will identify the broader impacts label-free optical technologies can have including implications in developing countries and opportunities in brain research. They will also be discussing the ultimate goal of identifying and prioritizing a path forward – so stay tuned for more details tomorrow. The hosts also plan to work with participants to publish a white-paper following the event to identify those recommendations.
